Water treatment remains a critical aspect of environmental management and public health. Hard water, characterized by high mineral content, poses unique challenges. The presence of calcium and magnesium can lead to scale formation and efficiency loss in water systems. According to a report by the Water Quality Association, 85% of U.S. water is classified as hard. This highlights the widespread need for effective treatment solutions.
A Hard Water Dehydrating Agent plays a vital role in addressing these challenges. It helps reduce mineral buildup and enhances water efficiency. The American Society of Civil Engineers emphasizes that proper water treatment can save municipalities up to 25% in operational costs. This statistic underscores the economic benefits of employing effective dehydration solutions.
However, not all treatments are flawless. Some agents can have limitations or require frequent adjustments. Industry professionals must critically evaluate their effectiveness. This constant need for improvement in water treatment methods reveals the complexity of managing hard water. The pursuit of optimal solutions, including the use of a Hard Water Dehydrating Agent, is essential for enhancing water quality and sustainability.


Hard water is defined as water containing high levels of minerals, primarily calcium and magnesium. These minerals can impact water quality significantly. According to the U.S. Geological Survey, approximately 85% of the United States has hard water. The presence of these minerals leads to issues such as scale buildup in plumbing and ineffective soap lathering. In homes, this often manifests as dingy laundry or cloudy dishes.
Understanding the characteristics of hard water is crucial for effective treatment. Scaling can reduce the lifespan of water heaters and appliances by up to 30%, according to estimates by the Water Quality Association. Additionally, hard water can contribute to the increased use of detergents, impacting overall cleaning efficiency. This not only affects household tasks but also poses challenges in industrial settings where water treatment is essential.
A comprehensive approach to treating hard water is necessary. With rising water usage and demand, the need for effective dehydration agents becomes more pronounced. These agents help manage mineral levels, ensuring water is not only safe but also effective for daily use. However, the challenge remains: how to strike a balance between mineral removal and maintaining essential water characteristics.
Dehydrating agents play a crucial role in water treatment processes, particularly when dealing with hard water. Hard water contains high levels of minerals like calcium and magnesium. These minerals can cause scaling and reduce the efficiency of treatment systems. By using dehydrating agents, operators can effectively remove excess minerals from water sources.
The mechanism of dehydrating agents involves binding with water molecules, which facilitates the removal of unwanted minerals. This not only improves water quality but also enhances the longevity of the treatment system. However, the choice of dehydrating agent is not straightforward. Factors such as water composition and treatment goals must be considered. Sometimes, the selected agent may not yield the expected results, leading to inefficiencies that warrant further investigation.
While dehydrating agents provide many benefits, their use comes with challenges. For instance, over-reliance on these substances can lead to unexpected changes in water characteristics. Operators must regularly test water to ensure optimal results. Continuous learning and adaptation in choosing the right agents enhance the overall effectiveness of water treatment processes.
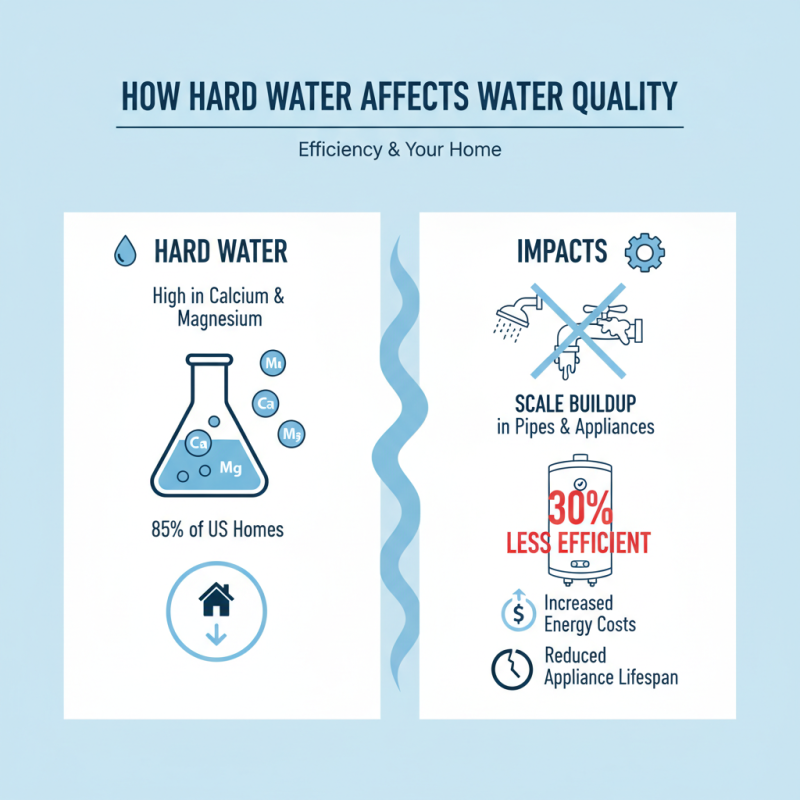
Hard water, characterized by high levels of calcium and magnesium, significantly affects water quality. According to the Water Quality Association, about 85% of U.S. homes have hard water. This hardness can lead to scale buildup in plumbing and appliances, reducing their effectiveness over time. A study from the American Water Works Association indicates that hard water can reduce the efficiency of water heaters by up to 30%. This drop in efficiency often leads to increased energy costs and premature equipment failure.
In addition, hard water complicates the effectiveness of water treatment chemicals. When hardness minerals are present, they can interfere with the action of disinfectants like chlorine. This interference can limit bacterial kill rates, posing potential health risks. Research highlights that water with high hardness levels requires about 20% more disinfectant to achieve the same water quality standards, which may strain both resources and budgets. With the combination of these factors, it becomes crucial to utilize hard water dehydrating agents in treatment processes. These agents help manage hardness levels, improving overall efficiency and ensuring safe drinking water.
Hard water poses significant challenges in water treatment. High mineral content, such as calcium and magnesium, can impact water quality. Dehydrating agents play a crucial role in managing these minerals effectively. According to industry reports, about 85% of homes in the U.S. have hard water. This widespread issue necessitates efficient solutions to improve water quality.
Using dehydrating agents brings multiple benefits. They help in minimizing scale build-up, which is detrimental to plumbing systems. Effective use of these agents can reduce maintenance costs significantly, saving homeowners up to 30% on repairs. Additionally, they enhance the efficiency of water systems, leading to better water flow and heating efficiency. The National Association of Home Builders estimates that softening hard water can improve energy efficiency by 15-20%.
Despite the advantages, the application of dehydrating agents isn’t without limitations. Some agents may not fully eliminate mineral deposits. The quality and composition of the treatment chemicals matter greatly. It’s essential to conduct water testing for optimal results. Striking the right balance in treatment choices is crucial for effective hard water management.
Dehydrating agents play a crucial role in water treatment processes. They effectively reduce hardness in water, making it more suitable for various applications. One notable case involved a municipality facing high mineral content in its water supply. By implementing a dehydrating agent, the city improved water quality significantly, leading to enhanced operational efficiency for local industries.
Another case study highlights the impact of dehydrating agents in agricultural settings. A farming community struggled with soil salinity, which affected crop yields. By treating irrigation water with dehydrating agents, farmers managed to lower sodium levels. This resulted in healthier crops and higher productivity. However, not every attempt yielded perfect results. Some farmers experienced initial setbacks, prompting a reevaluation of treatment methods.
The experience of using dehydrating agents conveys valuable lessons. Challenges can arise during implementation, and not all approaches work universally. It’s important to analyze specific conditions to achieve optimal results in water treatment. Understanding the local environment and water characteristics is crucial for success.