Nickel plating is a widespread method for enhancing metal surfaces. A nickel plating protective agent is vital in ensuring durability and resilience. According to a report by Global Market Insights, the nickel plating industry is expected to surpass $32 billion by 2026. This growth reflects its importance in sectors like automotive and electronics.
Dr. Emily Watson, an esteemed expert in surface engineering, emphasizes the significance of nickel plating protective agents. "Investing in proper nickel plating methods can significantly extend the lifespan of metal components," she states. This highlights the critical role these agents play in corrosion resistance and wear protection.
Despite its benefits, the nickel plating process is not without challenges. Proper application is crucial, as uneven layers can lead to defects. Moreover, environmental concerns arise from nickel waste. Therefore, continuous research and innovation in nickel plating protective agents remain essential for sustainable growth.

Nickel plating is a versatile process used to enhance metal surfaces. It involves applying a thin layer of nickel onto a metal substrate. This layer provides various benefits such as corrosion resistance, improved appearance, and increased surface hardness. The process is commonly used in industries like automotive, electronics, and manufacturing.
The nickel plating process starts with surface preparation. The metal items are cleaned to remove contaminants. This ensures that the nickel adheres well. Next, a nickel solution is used to deposit material onto the surfaces. The plating thickness can vary based on the application needs. However, achieving uniform coverage can be challenging. Areas with complex shapes may receive inconsistent layers.
After plating, a protective agent is often applied to enhance durability. This agent can improve resistance to scratches and environmental factors. It’s important to monitor the quality throughout the process. Any inconsistencies can lead to reduced effectiveness. Regular testing is required to maintain desired standards.
Nickel plating is widely used to enhance the corrosion resistance and aesthetic appeal of metal surfaces. A key component in this process is the protective agent. These agents reduce the formation of micro-corrosion during and after plating. Their role ensures a smoother finish and stronger adhesion of the nickel layer. The use of protective agents can lead to improved durability, with studies showing they can increase corrosion resistance by up to 30%.
However, not all protective agents are created equal. Some may enhance plating processes while others might unintentionally introduce defects. Inspecting the quality and compatibility of protective agents is crucial. Data from industry reports indicate that up to 15% of plating failures stem from inadequate agent selection. It’s essential to evaluate these agents thoroughly to prevent costly rework.
Tips: Always test new protective agents in small batches. Monitoring the results can help identify any negative interactions. Staying informed about recent advancements in protective technology can also provide valuable insights. A well-planned approach to using protective agents can significantly improve the outcome of nickel plating.
| Property | Description |
|---|---|
| Function | Provides corrosion resistance and enhances the durability of nickel plating. |
| Composition | Typically a mixture of organic and inorganic compounds. |
| Application Method | Applied during the plating process to form a protective layer. |
| Benefits | Improves adhesion, reduces stress, and enhances the finish of nickel plating. |
| Environmental Impact | Many modern formulations are designed to be more environmentally friendly. |
| Common Uses | Used in automotive, aerospace, and electronic components for enhanced protection. |
Nickel plating is widely used in various industries to enhance the lifespan of metals. A protective agent plays a crucial role in this process. These agents can improve corrosion resistance and provide a hard surface finish. By making the surface tougher, they help prevent wear and tear over time. This is essential for components that face harsh environments.
Durability is key when it comes to metal parts. Nickel plating protective agents create a barrier that minimizes damage from moisture and chemicals. They fill in small imperfections on the metal surface, making it smoother and reducing friction. This reduction in friction can extend the life of mechanical parts significantly.
*Tip: Always consider the thickness of the nickel layer. Too thin may not provide adequate protection, while too thick can lead to cracking. Balance is crucial for optimal results.*
When choosing a protective agent, evaluation is vital. Not all agents perform equally. Testing various agents can provide insights into their performance. This helps determine which one best suits the application’s needs.
*Tip: Regular maintenance and inspections are essential. Even with protective agents, wear may occur over time. Keeping an eye on the condition helps in timely interventions.*
Nickel plating protective agents are used to enhance corrosion resistance and improve surface durability. Various application methods exist for these agents, each with its specific requirements and benefits. The most common method involves electroplating, where a nickel salt solution is used. This process requires precise control of current and temperature to achieve optimal results.
Another method is immersion plating. In this approach, the item to be plated is submerged in a nickel-containing solution. It offers uniform coverage but requires careful handling to avoid pooling or uneven application. Spray plating is also gaining traction. It allows for localized application, which can be beneficial in repairing small areas.
Challenges arise with each method. For instance, maintaining consistent thickness can be tricky with immersion plating. Electroplating may also face issues like hydrogen embrittlement if not managed properly. These considerations are crucial for achieving the desired outcomes and ensuring long-term performance. Understanding these nuances can significantly impact the quality and effectiveness of nickel plating applications.
This chart illustrates the usage frequency of various nickel plating protective agents. Electroplating is the most commonly used method, accounting for a significant portion, followed by Physical Vapor Deposition (PVD) and Chemical Vapor Deposition (CVD). Electroless plating, while less common, still plays a vital role in specific applications.
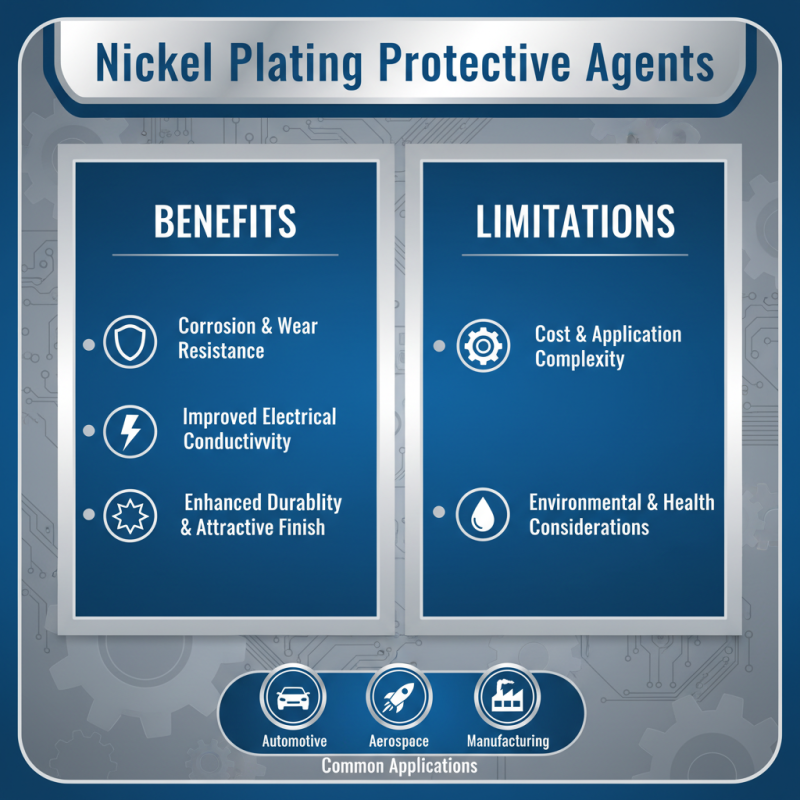
Nickel plating protective agents provide a layer of defense against corrosion and wear. They enhance the durability of various metal surfaces, making them suitable for diverse applications. The benefits of using these agents are significant. They help improve electrical conductivity and provide an attractive finish. Nickel plating is often used in automotive, aerospace, and manufacturing sectors.
However, there are limitations to consider. The thickness of the nickel layer can affect its protective capabilities. Too thin a layer may not provide adequate protection. Additionally, the process can be expensive and time-consuming. Environmental concerns regarding nickel toxicity also raise questions about sustainability. These factors require careful evaluation before proceeding with nickel plating applications.
Choosing the right nickel plating protective agent involves balancing these benefits and limitations. It is essential to assess the specific needs of each project. Experience with different plating techniques can inform better decision-making. Awareness of potential issues can help in selecting appropriate materials. Conducting thorough research and consulting experts are critical steps in this process.